Nobel Prize in Chemistry for the development of lithium-ion batteries

Researchers Stanley Whittingham, John Goodenough and Akira Yoshino will receive the 2019 Nobel Prize in Chemistry for the development of lithium-ion batteries. The Nobel Foundation has announced that lithium-ion batteries have revolutionized our lives. Thanks to them, they have opened the way to a society without cables and without fossil fuels.
Lithium-ion batteries are currently used from mobile phones to electric vehicles. They are light, rechargeable and powerful. In this way, there are many advances that have brought to society: first, all portable electronics are powered with them in the world; they have also served to develop powerful electric cars; and, in addition, they have served to store energy from renewable sources, such as the sun and wind.In the opinion of the Foundation, all these areas were necessary for this type of powerful batteries, capable of storing large amounts of energy, to be really successful.
The instability of lithium, its strength
Lithium is a metal. It is a very unstable element - it must be kept in oil so that it does not react with the air - with a single electron around the nucleus, which tends to leave the lithium to go to another atom. This is how lithium ions are formed, when they are positively charged, and they are much more stable than lithium itself.
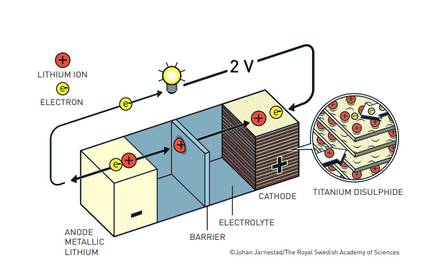
This unstable reactivity of lithium is, however, its strength.Stanley Whittingham took advantage of this tendency of lithium to release the electron by creating the first functional lithium battery. Growing concern about oil shortages prompted the oil industry to enter energy research. Exxon welcomed some of the most important researchers in the field of energy, offering a lot of money and research freedom. Among them, the English researcher Stanley Whittingham. As he researched superconducting materials, he created the first battery with titanium disulfide cathode and metallic lithium anode. He obtained a high-potential battery, which generated two volts, but was not stable.
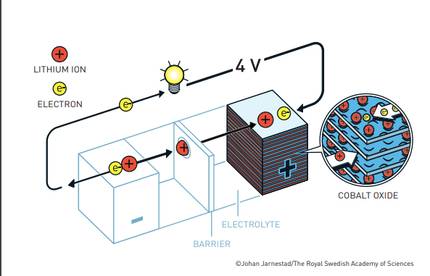
In 1980, John Goodenough doubled the battery potential by finding conditions that made the battery much more powerful. It envisaged greater battery potential by replacing metal sulfide with metal oxide. And he showed that with cobalt oxide, interspersing lithium ion, the battery could generate up to four volts.
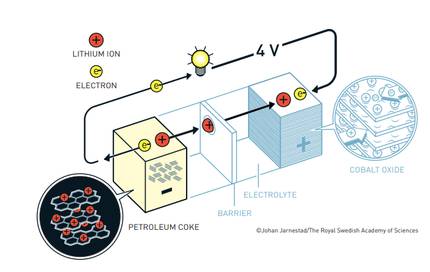
In 1985, Akira Yoshino replaced pure lithium from the battery with lithium ion. The advantage of lithium-ion batteries is that they are not based on chemical reactions that break down the electrodes, but on lithium-ion that move between the anode and the cathode. Its result was a very light and resistant battery, which could be recharged hundreds of times, without deteriorating its performance. So it got the battery really useful and saleable.
The Nobel Foundation has recognized that the three researchers have contributed much to society. They claim to have created a "rechargeable world."